The Post-Merger Watson™ Problem: How to Phase Out Watson LIMS™ Cost-Effectively
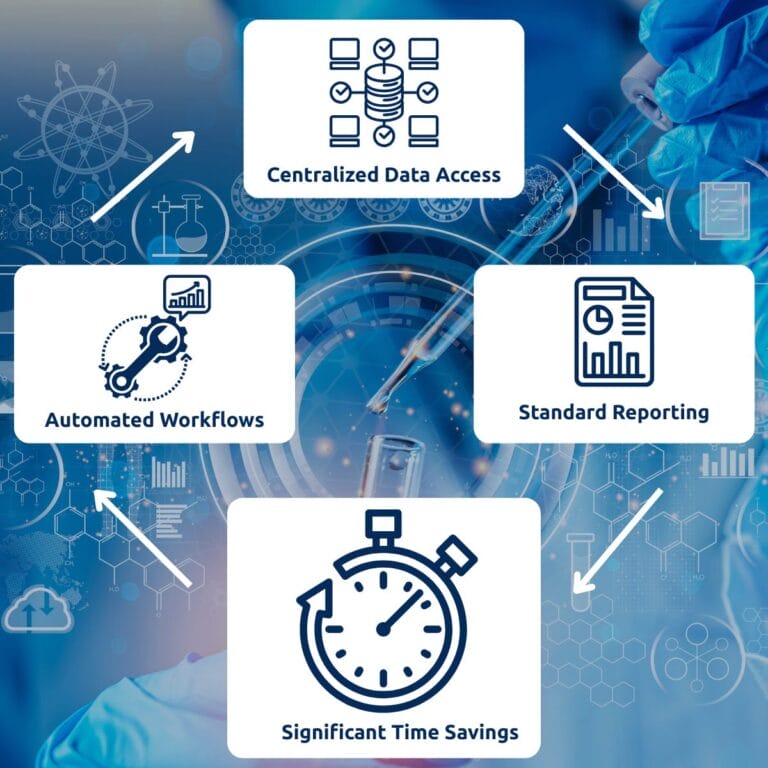
Blog The Post-Merger Watson LIMS™ Problem: A Cost-Effective Path to Decommissioning A CFO/CIO guide to decommissioning legacy bioanalytical LIMS without compliance risk. Post-merger application rationalization moves quickly until it hits a GxP-regulated system. Both sides of a pharmaceutical M&A often run Watson™ LIMS, and neither environment appears safe to decommission without creating a compliance gap….