up to data Blog
Discover
Innovation Impulses
and
Latest Insights
up to data Blog
Discover Innovation Impulses
and Latest Insights

Explore how advanced technology is reshaping clinical trial data management.
The up to data blog examines industry trends in laboratory technology, artificial intelligence, data automation, research efficiency, and quality improvement. Follow insights on how emerging technologies are driving precision medicine forward and shortening the pathway from research to effective therapies.
-
Current
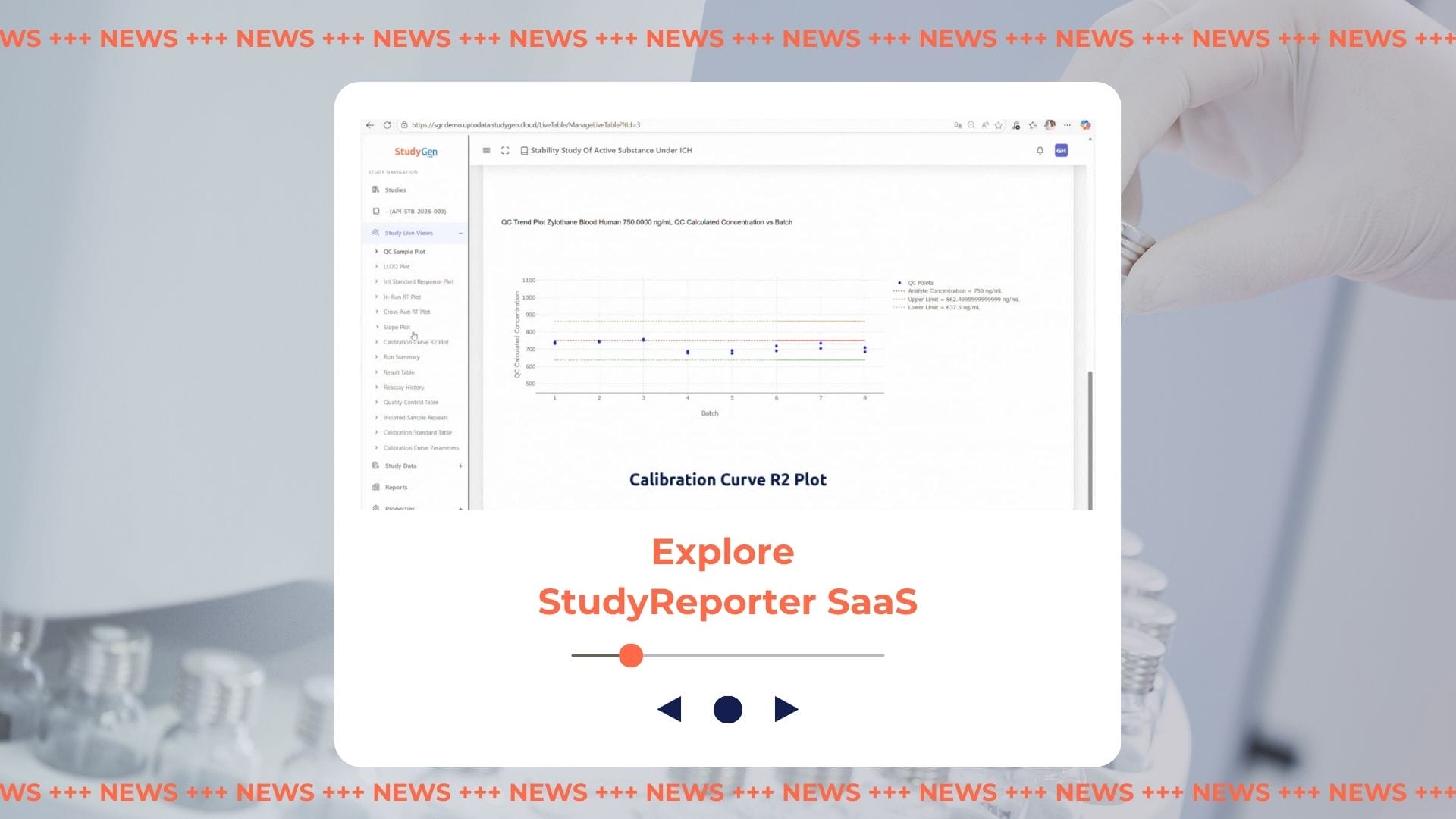
Beyond Manual Reporting | Sneak Peak StudyReporter
03/13/2026Blog Beyond Manual Reporting: A Sneak Peek into the Future of Automated Study Data Reporting Discover in our exclusive sneak peek video how StudyReporter automates the creation of GxP-compliant study reports and reduces manual effort by up to 70%. Develop your study reporting workflow from source to submission—efficiently, securely, and with maximum data integrity. Watch… -
Knowledge
The Post-Merger Watson™ Problem: How to Phase Out Watson LIMS™ Cost-Effectively
05/07/2026Blog The Post-Merger Watson LIMS™ Problem: A Cost-Effective Path to Decommissioning A CFO/CIO guide to decommissioning legacy bioanalytical LIMS without compliance risk. Post-merger application rationalization moves quickly until it hits a GxP-regulated system. Both sides of a pharmaceutical M&A often run Watson™ LIMS, and neither environment appears safe to decommission without creating a compliance gap…. -
Knowledge
Long-Term Retention of Bioanalytival Watson LIMS studies: A Compliance-First Perspective
05/07/2026Blog Long-Term Retention of Bioanalytical Watson LIMS™ Studies: A Compliance-First Perspective Most bioanalytical labs running Watson LIMS™ have never made a conscious decision about archiving. They have simply deferred. What 21 CFR Part 11 and EU GMP Annex 11 actually require, and why format independence is a regulatory posture, not an IT preference. Executive Summary The majority… -
Knowledge
Four Dimensions of Time Savings in GxP Bioanalytical Study Reporting
04/16/2026Blog Four Dimensions of Time Savings in GxP Bioanalytical Study Reporting With rising expectations under ICH M10, FDA Part 11, and EMA guidelines, bioanalytical labs face growing pressure: more data, more documentation, and less room for error. Manual reporting still risks transcription mistakes, slows QC cycles, and complicates inspection readiness.This article highlights four dimensions where automation drives measurable… -
Knowledge
Automated Reporting for GxP Labs
02/13/2026Blog From Raw Data to Decision: Why Automated Reporting is Essential for GxP Labs Bioanalytical reporting involves converting raw analytical data from validated methods into structured, submission-ready documentation for regulatory purposes or sponsor decision-making. This process links each sample, chromatogram, or assay signal to the final submitted concentration or pharmacokinetic (PK) parameter, enabling traceability, and… -
Knowledge
CRO-Sponsor Collaboration: Digital Risk Mitigation
08/14/2025Blog Optimizing CRO-Sponsor Collaboration: Mitigating Risks in Bioanalytical Study Reporting Manual processes in bioanalytical studies jeopardise efficiency, data integrity, and compliance – especially under the strict ICH M10 requirements. This article shows how digital platforms like StudyGen 360 leverage centralised data access, real-time collaboration, and automated workflows to minimise errors, accelerate studies, and future-proof CRO–sponsor relationships. Why… -
Knowledge
Context-First AI Approach for CROs | Multi-Run Assay Control
06/25/2025Blog The Critical Role of Multi-Run Assay Control in Bioanalytical Research: A Context-First AI Approach for Contract Research Organizations (CROs) In bioanalytical research, precise and reliable data is of the utmost importance. This article examines the necessity of robust quality control (QC) systems with a specific focus on multi-run assay control procedures for Contract Research… -
Knowledge
AI in GxP Environments: Context-First Approach
03/26/2025Blog Leveraging AI in GxP Environments: A Context-First Approach to Bioanalytical Data Management In regulated bioanalytical laboratories, the promise of artificial intelligence has often seemed at odds with the stringent requirements of GxP compliance. Traditional approaches to AI implementation face significant hurdles: data privacy concerns, validation challenges, and the complexity of maintaining compliance when using… -
Success Story
Digital Transformation INQA Success Story
03/24/2025Blog The Challenge of Digital Transformation: How up to data GmbH Shapes Future-oriented Change The digital transformation and the changes associated represent a particular challenge for medium-sized companies. up to data GmbH meets this challenge with a sophisticated concept and professional support through the INQA coaching program, an initiative coordinated by the Federal Ministry of… -
Knowledge
Standardized Multi-Run Assay Control Guide Recording
03/10/2025Blog Enabling Standardized Multi-Run Assay Control: Key Insights for Bioanalytical Labs In an increasingly complex regulatory environment, bioanalytical laboratories face growing challenges in maintaining consistent assay performance across long-running studies. A recent webinar led by Norbert Bittner, CEO of up to data, provided comprehensive insights into implementing standardized multi-run assay-controls and highlighted effective strategies for…