Blog
The Post-Merger Watson™ Problem: How to Phase Out Watson LIMS™ Cost-Effectively
A CFO/CIO guide to decommissioning legacy bioanalytical LIMS without compliance risk. Post-merger application rationalization moves quickly — until it hits a GxP system. Both sides of a pharmaceutical M&A often run Watson LIMS™, and neither environment appears safe to turn off without creating a compliance gap. This article challenges that assumption: the regulatory requirement is for data access, not system operation — and the costs of leaving a frozen Watson™ instance running compound with every year of inaction.

Executive Summary
If your organization has emerged from an M&A deal with more than one Thermo Watson LIMS™ environment in its portfolio, you face a familiar impasse: neither can be simply switched off. The studies they contain are subject to GxP data-retention requirements of 15 years or more, and your scientific teams — reasonably — want data access guaranteed.
Keeping Watson™ running is not the only way to satisfy that requirement — but it is typically the most expensive approach. The annual cost of a single legacy Watson™ environment runs to approximately $150,000–$160,000 per site, a figure that compounds as Oracle support escalates and platform end-of-life events approach. A one-time decommissioning investment that pays for itself inside Year 2 converts every completed study into a regulator-ready, vendor-independent archive and eliminates all ongoing Watson™ costs from Year 2 onward. Payback comes inside Year 2. Over a standard 15-year retention horizon, the net saving is approximately $3 million per site — roughly 17× the initial investment.
The Situation You’re In
Post-merger application rationalization tends to move quickly until it reaches GxP systems, where progress typically stalls.
Watson LIMS™ is a common example of this dynamic. Both sides of many pharmaceutical acquisitions run Watson™. Neither environment can be turned off without, at first glance, creating a compliance gap. So both remain on the IT estate, consuming budget, absorbing FTE bandwidth, and complicating the integration timeline.
This brief is for the IT and finance leaders responsible for resolving that gap. The core argument is straightforward: the compliance requirement applies to data access, not to system operation. Watson™ was designed as an operational system, not a long-term archive. Once a study is complete, the system continues to generate infrastructure costs without generating scientific output.
It is also relevant regardless of how long ago the acquisition occurred. The integration window creates organizational momentum to address application rationalization — but the economic argument for decommissioning a legacy Watson™ estate does not expire with it. An organization that has been running an inherited Watson™ environment for three or five years faces the same cost drivers, the same regulatory retention obligation, and the same decommissioning path. The costs incurred to date cannot be recovered; the costs from this point forward are entirely avoidable.
Why Watson™ Is Still Running: The “Just in Case” Problem
Legacy Watson™ environments in post-merger estates share a defining characteristic: they are running, but producing nothing. No new studies are being entered. No configuration changes are being made. Watson™ cannot be extended, customized, or evolved for new requirements — the system is, in practice, frozen. The sole remaining use case is occasional data retrieval: a response to an FDA inspection query, a sponsor request, or an internal audit.
That retrieval scenario is real and legitimate. Your scientists and QA teams are right to want that access guaranteed. But the assumption that Watson™ itself must remain operational to provide it is where the analysis breaks down.
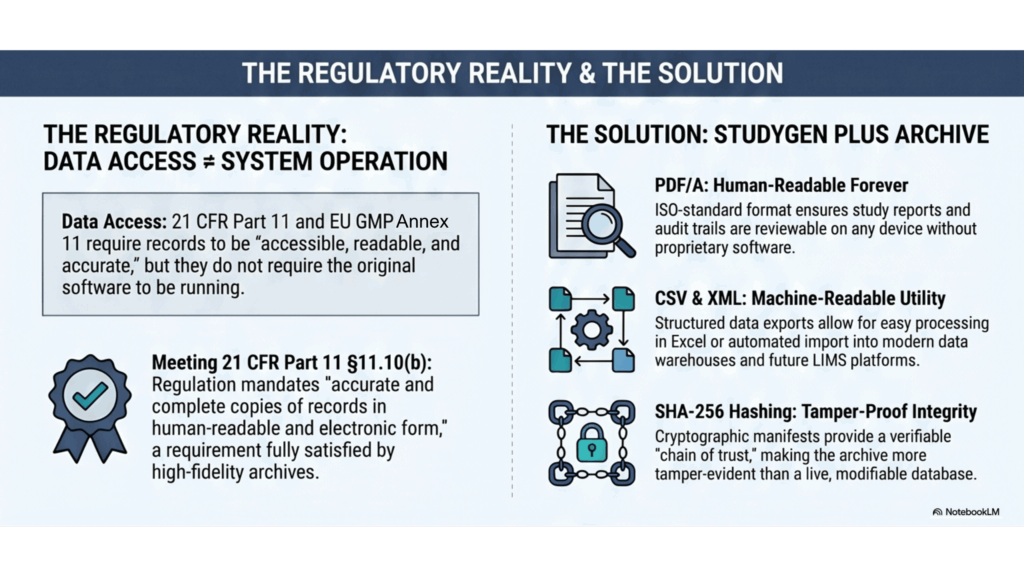
The regulatory requirements — 21 CFR Part 11 §11.10(b) and EU GMP Annex 11 §7 — require human-readable, accurate, and complete copies of records to be preserved throughout the retention period. Not for the originating system. An archived study, exported as structured PDF/A documents with full CSV and XML datasets and cryptographic integrity verification, satisfies both standards fully — without a single Watson™ component running.
| Boehringer Ingelheim: From the Same Starting Point Boehringer Ingelheim — one of the world’s largest pharmaceutical companies — started from the same position. Their scientific and QA teams had the same concerns about data accessibility after decommissioning. After a structured archiving process using the StudyReporter Archiving Module, they successfully retired their Watson™ environment. Every study remains fully accessible, audit-ready, and compliant with applicable regulations. Watson™ is gone. |
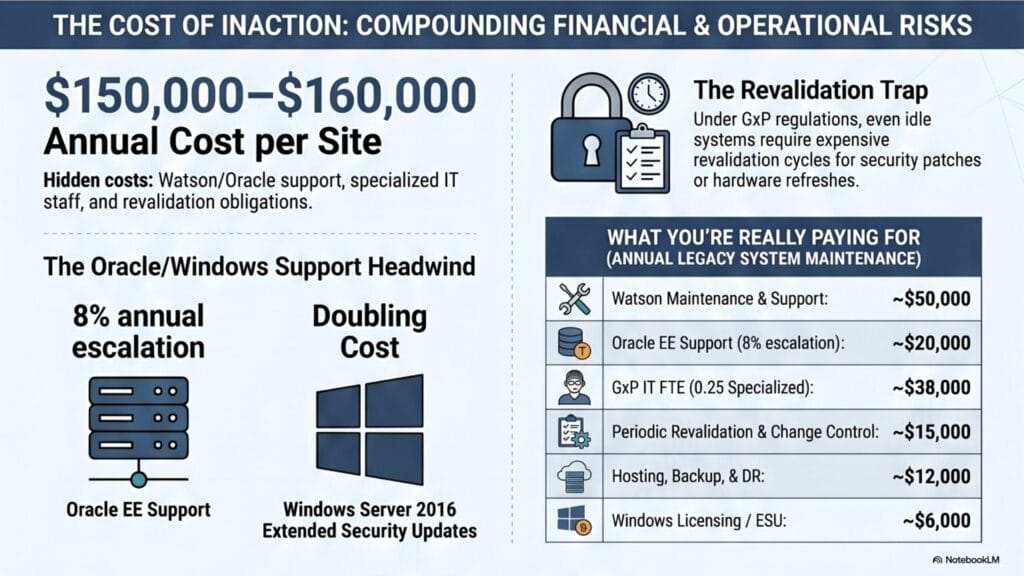
What You’re Really Paying For
The cost of keeping a legacy Watson™ environment alive is distributed across several line items — none of which generate new scientific value. The table below shows a conservative ballpark breakdown for a typical mid-sized Watson™ deployment.
| Cost Item | ~USD / Year / Site |
| Watson™ maintenance & support | ~$50,000 |
| Oracle EE support (escalating ~8%/yr) | ~$20,000 — rising |
| GxP IT FTE (0.25 FTE, specialized) | ~$38,000 |
| Periodic revalidation & change control | ~$15,000 |
| DR/hosting/backup | ~$12,000 |
| Windows Server licensing / ESU | ~$6,000 |
| Total per site | ~$150,000–$160,000 |
Four Headwinds Making This Worse Each Year
Four independent factors drive the annual cost upward regardless of how actively the system is used.
Oracle Enterprise Edition support escalation
Watson™ requires Oracle™ Database Enterprise Edition — list price approximately $47,500 per processor, with annual support at 22% of the net license. Oracle’s support escalation policy runs at approximately 8% per year, compounding indefinitely. There is no negotiated path to lower support-only pricing once the licenses are in place.
Windows Server 2016 end of support — January 2027
Microsoft ends mainstream support for Windows Server 2016 in January 2027. Extended Security Updates (ESU) are available, but the price doubles each year. Migrating to a newer Windows version requires a full GxP revalidation of the Watson™ stack — a time-consuming and costly exercise for a system that is no longer producing data.
GxP IT skills scarcity
Maintaining a validated GxP system — running Oracle EE, managing IQ/OQ revalidation cycles, handling validated change control — requires a specialized and increasingly scarce skill set. The FTE allocation for a legacy Watson™ stack that no one actively uses is a disproportionate and growing drain on a resource your IT organization likely needs elsewhere.
Revalidation obligation regardless of activity
Under GxP regulations, any change to the underlying platform — a security patch, a hardware refresh, an OS update — triggers a revalidation obligation regardless of whether the system is in active use. A frozen, idle system on aging infrastructure requires exactly the same validated change management discipline as a fully productive one.
The Actual Requirement: Data Access, Not Watson™ Access
The compliance rationale for keeping Watson™ running often rests on an imprecise reading of what the applicable regulations actually require.
21 CFR Part 11 §11.10(b) requires the ability to generate “accurate and complete copies of records in both human-readable and electronic form”. EU GMP Annex 11 §7 requires that “data must remain accessible, readable and accurate throughout the retention period”. Neither regulation specifies the originating system. Neither requires Watson™ to be operational.
The StudyReporter Archiving Module satisfies both standards directly. For every completed Watson™ study, it generates:
- PDF/A documents — ISO-standard long-term archival format, human-readable on any device, without any proprietary software, forever.
- CSV and XML datasets — every analytical result, calibration, sample, regression, and audit trail in structured, machine-readable form, compatible with any data warehouse or future LIMS platform.
- A cryptographic integrity manifest — a SHA hash for every output file, plus a “hash of hashes” manifest logged in the processing protocol. Any post-archive modification to any byte is immediately and provably detectable.
The archive is, in an important sense, more tamper-evident than Watson™ itself. Records in a running Oracle database can be modified. A signed, hash-verified flat-file archive cannot be silently altered. The archive format is also permanently vendor-independent: it requires no Watson™ license, no Oracle™ license, or any additional licenses — especially no proprietary viewer — now or in the future.
After archiving, the organization retains full regulatory utility while eliminating the ongoing infrastructure obligation entirely.
The Business Case
The following figures are based on the up to data ROI model, available in full upon request. All numbers are ballpark estimates; actual figures vary by Oracle footprint, FTE rate, and site configuration. For organizations with more than one Watson™ environment, the figures scale proportionally.
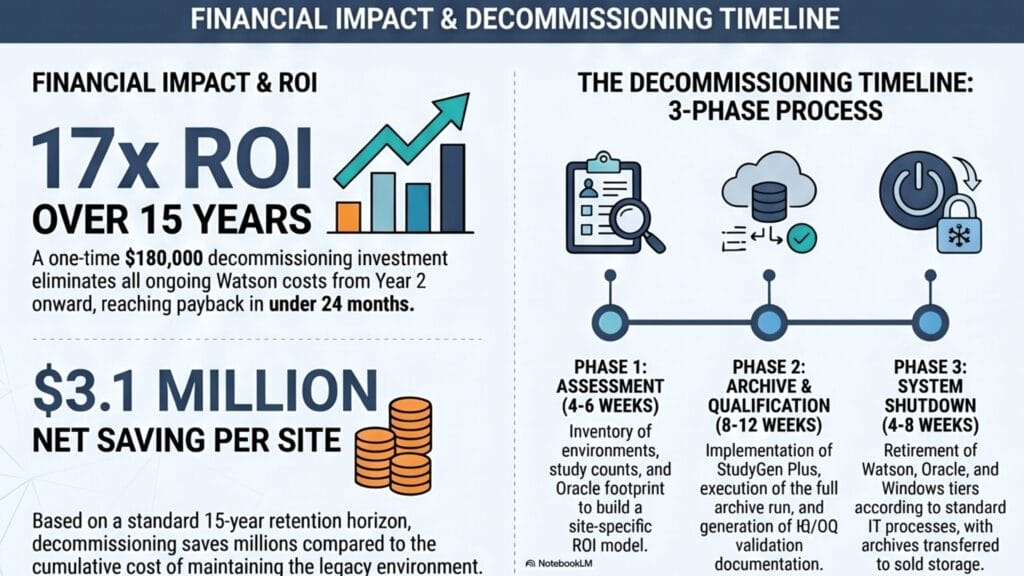
| Criteria | Per Site |
| One-time investment | ~$180,000 |
| Annual savings from Year 2 | ~$155,000/yr |
| Payback period | ~2 years |
| Net saving — 10 years | ~$1.8M |
| Net saving — 15 years (retention horizon) | ~$3.1M |
| ROI multiple (15-yr horizon) | ~17x |
The one-time investment covers license, implementation, validation documentation (IQ/OQ), and a complete archive run across all completed studies. There is no annual support fee, no subscription, and no ongoing vendor relationship after the archive run. The StudyReporter Archiving Module is itself decommissioned alongside Watson™.
Boehringer Ingelheim is in production with this solution. A leading Japanese top-20 pharmaceutical company has independently completed the same transition. Both validated the approach through their standard GxP qualification process.
What a Structured Exit Looks Like — Standard Project Overview
A Watson™ decommissioning project runs in three phases, with a total elapsed time of 4 to 6 months from project start to system shutdown. The following provides a high-level overview of a standard StudyPlus Archive project.
Phase 1 — Assessment (4–6 weeks)
Inventory of Watson™ environments, study counts, Oracle footprint, and integration dependencies. Output: a decommissioning scope document and a site-specific ROI model calibrated to your actual cost inputs.
Phase 2 — Archive Run and Qualification (8–12 weeks)
StudyReporter Archiving Module configured, IQ/OQ validation documentation prepared, full archive run executed across all completed studies, and integrity verification completed and documented. Output: validated study archive, processing protocol, and QA sign-off package.
Phase 3 — Decommissioning (4–8 weeks)
Watson™, Oracle™, and Windows tiers retired according to your standard IT decommissioning process. Archive transferred to cold storage. Ongoing archive maintenance cost: effectively zero.
Request a Decommissioning Assessment
A Decommissioning Assessment is a structured 30-minute conversation in which we review your specific Watson™ environment — number of instances, Oracle footprint, study volume — and produce a tailored cost-of-inaction analysis against the ROI model. No commitment required. The output is a document you can take directly to your integration review board. Contact us to schedule your assessment at info[at]uptodata.com.
Conclusion: The Decision That Pays for Itself
The economic case for decommissioning a legacy Watson™ environment does not require optimistic assumptions. It requires only an honest accounting of what the system currently costs — in Oracle support, Windows Server infrastructure, GxP IT bandwidth, and revalidation overhead — versus a one-time investment that permanently eliminates all of those line items.
The compliance concern that keeps most post-merger Watson™ instances running is legitimate. But it rests on a misreading of what the regulation actually requires. 21 CFR Part 11 and EU GMP Annex 11 require access to data, not system operation. A validated, hash-verified archive of PDF/A documents and structured CSV/XML datasets satisfies both standards in full — without a single Watson™ component running.
The integration window creates organizational momentum to act. But the argument does not expire with it. An organization that has been carrying an inherited Watson™ environment for three or five years faces the same cost drivers, the same retention obligation, and the same decommissioning path. The costs incurred to date cannot be recovered. The costs from this point forward are entirely avoidable.
Supporting the Internal Stakeholder Discussion
We offer a companion document — Decommissioning Watson™: Answers to the Questions Your QA and Lab Directors Will Ask — designed to help IT and finance leaders address the scientific and compliance concerns that typically arise when decommissioning is first proposed.
References
European Commission (2011). EU guidelines for good manufacturing practice for medicinal products for human and veterinary use: Annex 11, computerised systems. URL: https://health.ec.europa.eu/system/files/2016-11/annex11_01-2011_en_0.pdf.
European Medicines Agency & Pharmaceutical Inspection Co-operation Scheme (2025). Joint stakeholders consultation on the revision of Chapter 4 on Documentation, Annex 11 on Computerised Systems and on the new Annex 22 on Artificial Intelligence of the PIC/S and EU GMP Guides. PIC/S. URL: https://picscheme.org/en/news/joint-stakeholders-consultation-on-the-revision-of-chapter-4.
U.S. Food and Drug Administration (2026a). 21 CFR Part 58.195: Good laboratory practice for nonclinical laboratory studies — Record retention. Electronic Code of Federal Regulations. URL: https://www.ecfr.gov/current/title-21/chapter-I/subchapter-A/part-58/subpart-K/section-58.195.
U.S. Food and Drug Administration (2026b). 21 CFR Part 11: Electronic records; electronic signatures. Electronic Code of Federal Regulations. URL: https://www.ecfr.gov/current/title-21/chapter-I/subchapter-A/part-11.
Appendix: Model Assumptions
All financial figures in this brief are ballpark estimates based on publicly available pricing and conservative industry-typical configurations:
- Oracle Database EE list price: $47,500 per processor (Oracle Technology Price List, 2026). Annual support: 22% of net license. Support escalation: ~8% per year.
- Watson LIMS™ licensing and support: not publicly disclosed by Thermo Fisher. Figures use conservative industry estimates; replace with contracted rates during qualification.
- Windows Server ESU: Microsoft Extended Security Updates, price doubles annually after the end of mainstream support (January 2027 for Windows Server 2016).
- GxP IT FTE: 0.25 FTE at $150,000 fully loaded annual cost (US/EU GxP sysadmin). Adjust to local market rates.
- The one-time investment of ~$180,000 represents a typical scope of work, including licensing, implementation services, and IQ/OQ documentation. Actual pricing varies by environment and is available on request.
- Full ROI model: An adjustable Excel model with configurable inputs for all cost assumptions is available on request from up to data GmbH.
up to data has been supporting pharmaceutical and life sciences companies with automated laboratory processes for regulatory study data management for over 20 years. Our solutions eliminate data silos, implement secure automated data transfer processes, and reduce manual activities while ensuring full regulatory compliance.
This content might also be engaging for you!
-
Long-Term Retention of Bioanalytival Watson LIMS studies: A Compliance-First Perspective
Blog Long-Term Retention of Bioanalytical Watson LIMS™ Studies: A Compliance-First Perspective Most bioanalytical labs running Watson LIMS™ have never made a…
-
Four Dimensions of Time Savings in GxP Bioanalytical Study Reporting
Blog Four Dimensions of Time Savings in GxP Bioanalytical Study Reporting With rising expectations under ICH M10, FDA Part 11, and EMA…
-
Beyond Manual Reporting | Sneak Peak StudyReporter
Blog Beyond Manual Reporting: A Sneak Peek into the Future of Automated Study Data Reporting Discover in our exclusive…