Blog
Beyond Manual Reporting: A Sneak Peek into the Future of Automated Study Data Reporting
Discover in our exclusive sneak peek video how StudyReporter automates the creation of GxP-compliant study reports and reduces manual effort by up to 70%. Develop your study reporting workflow from source to submission—efficiently, securely, and with maximum data integrity.
Watch the Sneak Peek: Less Manual Work, Faster Results
Whether you’re a small research lab or a large pharmaceutical company, StudyReporter is built to grow with your business. No LIMS? No worries! The versatile platform can handle direct uploads of bioanalytical sample data and produce fully compliant MS Word reports in just minutes. To learn more about seamless integration with existing laboratory data systems like Watson LIMS™ or ELNs, please visit our solution page for Large & Small Molecules.
Watch the video below to see StudyReporter in action:
From Manual Bottlenecks to Intelligent Automation
In the fast-paced world of clinical trials, turning raw laboratory data into a final, regulatory-compliant report can cause delays. Manual data collection and formatting can take days, making results prone to human errors.
At StudyGen 360, we believe your focus should be on analysis rather than data compilation. That’s why we’re excited to give you an exclusive look at how StudyReporter, the evolution of our trusted iStudyReporter, is transforming the management of digital study data.
Why Leaders are Moving to StudyReporter by StudyGen 360
Trusted by 15 of the top 25 pharmaceutical and biotech companies, our platform provides more than automation — it delivers regulatory confidence.
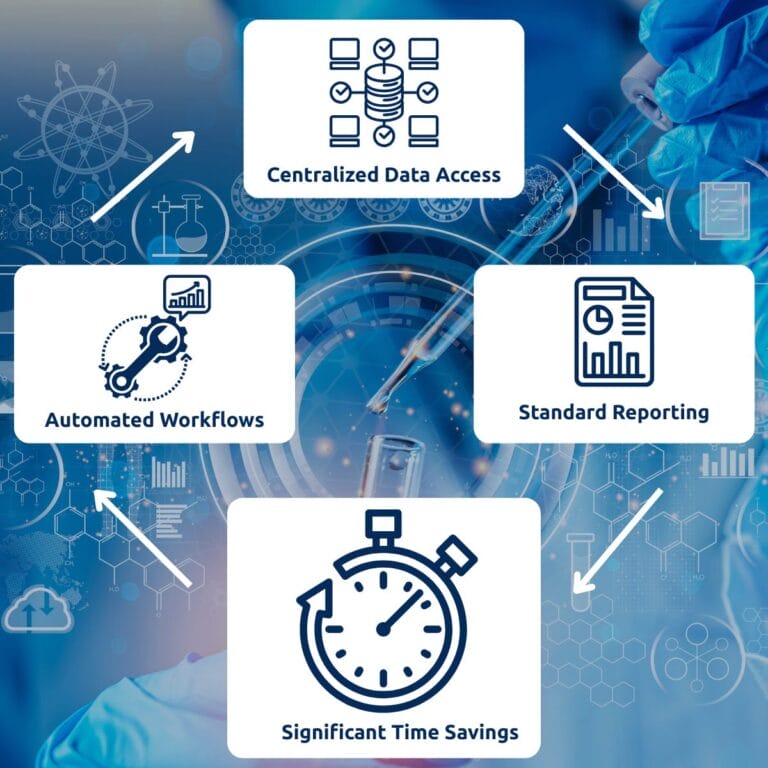
- 70% Time Savings: Reduce document creation time from days to minutes with automated data population.
- Compliance by Design: Built-in validation rules ensure compliance with GxP, FDA, and EMA, adhering strictly to 21 CFR Part 11 and ALCOA+ principles.
- Seamless Integration: Connect directly to Watson LIMS™, further Laboratory Management Systems (LIMS), or analytical instruments such as LC-MS and GC-MS.
- Real-Time Insights: Generate live tables and monitor study progress in real time, enabling proactive, fast decision-making.
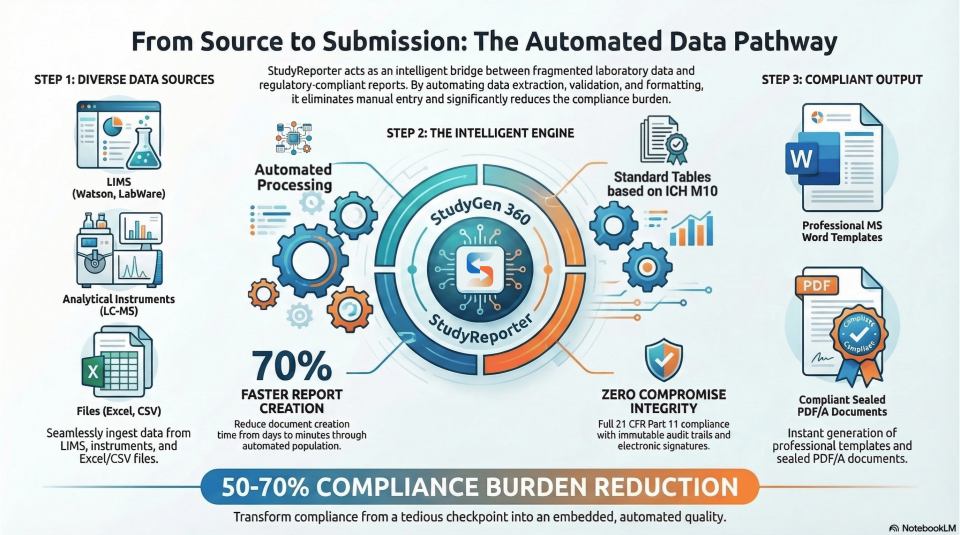
Visualizing the Workflow: From Source to Submission
To help you understand how StudyReporter fits into your ecosystem, here is a visualization of our automated data pathway.
Conclusion: Ready to Elevate Your Clinical Study Data Reporting?
Stop wasting time on manual data handling and start delivering measurable results more quickly. StudyReporter ensures every data point is fully traceable, from initial capture to the final report, ready for submission.
up to data has been supporting pharmaceutical and life sciences companies with automated laboratory processes for regulatory study data management for over 20 years. Our solutions eliminate data silos, implement secure automated data transfer processes, and reduce manual activities while ensuring full regulatory compliance.
This content might also be engaging for you!
-
Automated Reporting for GxP Labs
Blog From Raw Data to Decision: Why Automated Reporting is Essential for GxP Labs Bioanalytical reporting involves converting raw…
-
CRO-Sponsor Collaboration: Digital Risk Mitigation
Blog Optimizing CRO-Sponsor Collaboration: Mitigating Risks in Bioanalytical Study Reporting Manual processes in bioanalytical studies jeopardise efficiency, data integrity,…
-
Context-First AI Approach for CROs | Multi-Run Assay Control
Blog The Critical Role of Multi-Run Assay Control in Bioanalytical Research: A Context-First AI Approach for Contract Research Organizations…